AI-PLAX: AI-based Placenta Assessment and Examination
ID# 2019-4905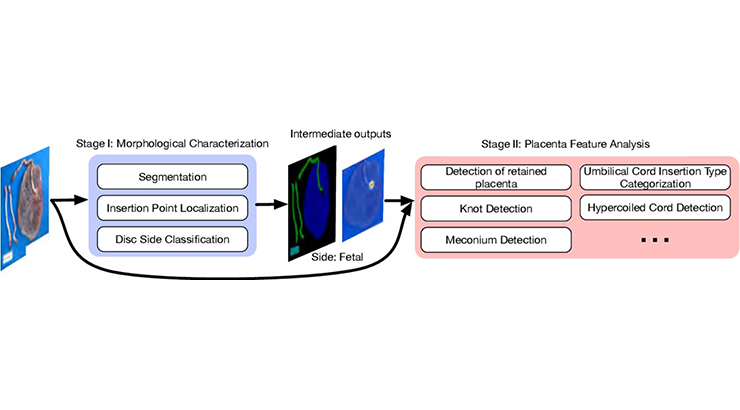
Technology Summary
The placenta is a complex organ with many critical roles during pregnancy. Generally, placentas are examined post-birth by the attending physician or midwife in a brief inspection that can be less than a minute. Placentas are sent for pathological exam only when it is considered necessary (based on the physician’s exam, maternal health, or fetal health) and resources are available. Research suggests that a significant number of placentas are either incorrectly sent for pathological exam when additional analysis was likely not needed or not sent for pathological exam even though recommended indications were present. Automated placenta analysis based on photographic imaging may allow more placentas to be examined, reduce the number of normal placentas sent for full pathological examination, allow for timely on-site initial diagnosis, and provide an objective morphological and pathological analysis. To automate the assessment of placentas, Penn State inventors have created a novel AI-based methodology and software that, using photographs of the placenta, extracts key visual features and predicts pathological diagnoses. Using large datasets of placenta photos and corresponding pathological records, Penn State inventors have demonstrated that the novel program can predict common pathological diagnoses, including abruption, meconium, hypercoiled cord, irregular shape, and marginal cord insertion, with up to 97% accuracy.
Application & Market Utility
Post-delivery analysis of the placenta is useful for evaluating health risks of both the mother and baby. In the U.S., however, only about 20% of placentas are assessed by pathology exams, and placental data is often missed in pregnancy research because of the additional time, cost, and expertise needed. A computer-based tool that can be used in any delivery setting at the time of birth to provide an immediate and comprehensive placental assessment would have the potential to not only to improve health care, but also to radically improve medical knowledge.
Next Steps
Seeking industry partners and/or licensees to drive clinical validation and commercialization.

