Dual-Switch Selection Gene Drives for Anticancer Therapy
ID# 2022-5426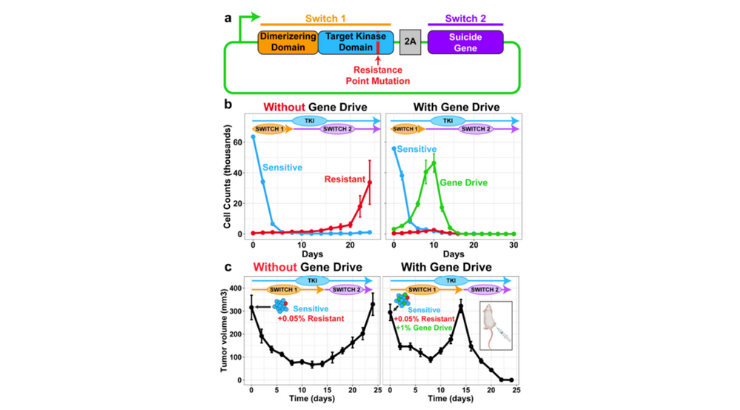
Technology Summary
Termed “dual-switch selection drives,” the selection drives induce temporary drug resistance to create a cell therapy that can engineer a tumor’s evolution in situ. The first switch senses the presence of a dimerizer molecule to create reversible drug resistance. Using the mathematical rules of biophysics and evolution, this cell therapy calculates a tunable fitness response to compete with pre-existing drug resistance variants in a tumor. This evolution guided cell therapy has a second switch that includes a suicide gene payload that takes effect until the selection drive cells comprise most of the tumor.
Application & Market Utility
The first target indication is non-small-cell-lung cancer (NSCLC), which has specific mutations in EGFR, RET, ALK, ROS1, MET and TRK that give rise to driver oncogenes and result in tumors with potent clinical responses to tyrosine kinase inhibitors. Higher resolution measurements have revealed that resistance mutations often pre-exist, and new drugs simply lead to new cycles of resistance. This resistance has a diversity of molecular mechanisms and few options. According to Nature Drug Discovery, the NSCLC drug market will nearly double to $48 billion by 2031.
Next Steps
Next steps would involve refining and optimizing the delivery of the dual-switch selection drives to effectively target and infiltrate the tumor microenvironment in non-small-cell lung cancer.

